Centers of Excellence
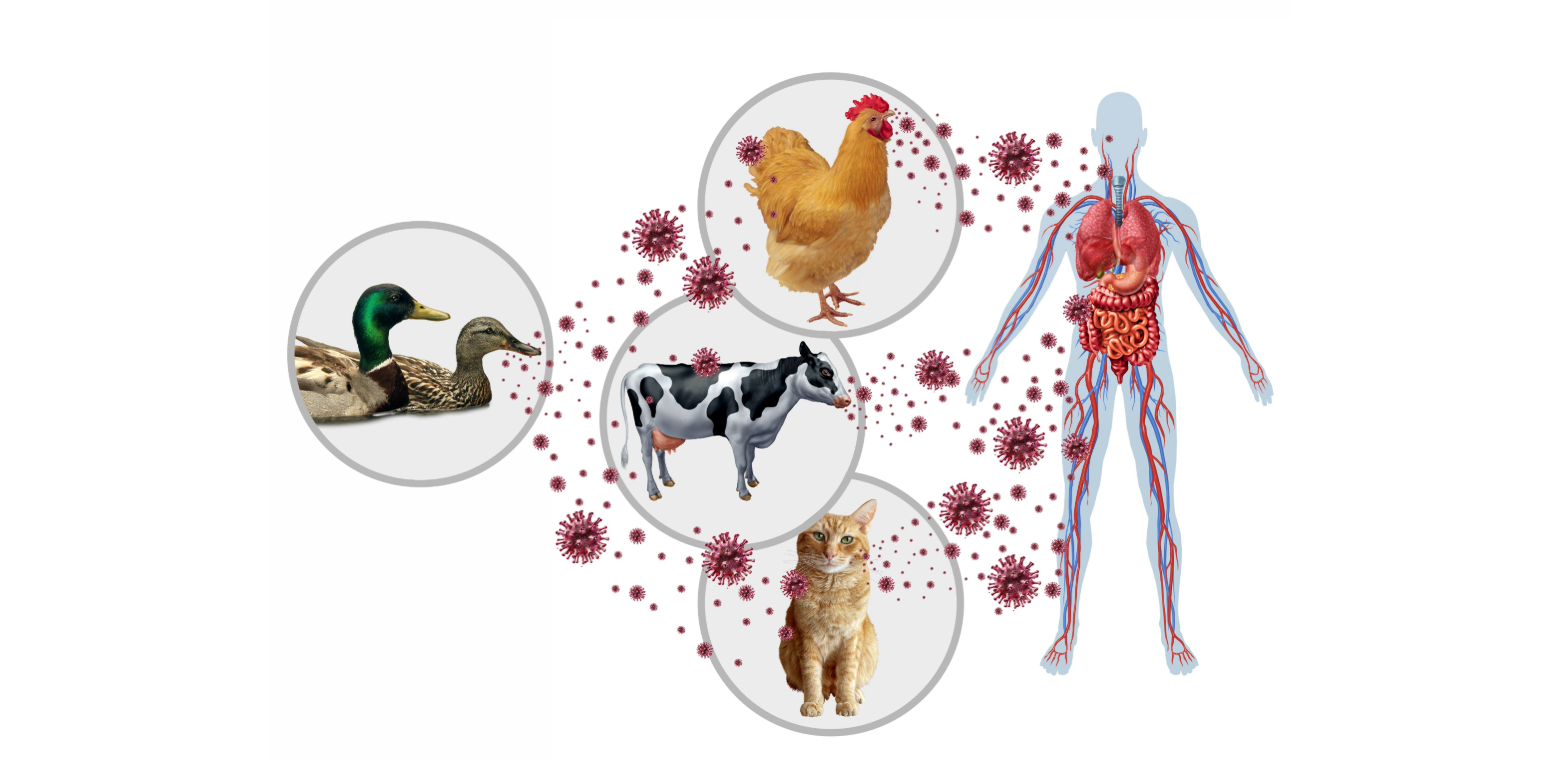
Academic Centers of Excellence & Affiliates conduct vital research in medical virology around the world. Together, they are dedicated to understanding, preventing, and responding to viral threats to human health, with a central focus on pandemic preparedness. Centers of Excellence serve as leading hubs with broad expertise in multiple viral areas, while Affiliates focus on specific areas of virology and are mentored by designated Centers of Excellence.
Centers & Affiliates
Countries

University of South Florida - GVN International Headquarters
12901 Bruce B. Downs Blvd.
MDC02
Tampa, FL 33612
- 1 of 92
 University of South Florida’s Institute for Translational Virology & Innovation - COE
University of South Florida’s Institute for Translational Virology & Innovation - COE
3814 Spectrum Blvd, #3101
Tampa, FL 33612
Center Director
Robert C. Gallo, MD
- 2 of 92


Harvard T.H. Chan School of Public Health/The Broad Institute - COE
665 Huntington Ave, Bldg 1, Room 706 Boston, MA 02115
Center Director
Pardis Sabeti, PhD, MD
- 3 of 92

Yale University - COE
- 4 of 92
Icahn School of Medicine at Mount Sinai - COE
One Gustave L. Levy Place, Box 1124, New York, NY 10029
Center Director
Peter Palese, PhD
- 5 of 92

Institute of Human Virology
725 W Lombard St, Baltimore, MD 21201
- 6 of 92

Johns Hopkins Bloomberg School of Public Health - COE
615 N Wolfe St, Baltimore, MD 21205
- 7 of 92
University of Rochester Medical Center, School of Medicine and Dentistry - COE
School of Medicine and Dentistry, 601 Elmwood Avenue, Box 609, Rochester, NY 14642
Center Director
David Topham, PI, URMC
- 8 of 92

University of Buffalo HIV and HCV Clinical Pharmacology Laboratory in Bioinformatics and Life Sciences - COE
701 Ellicott St, Buffalo, NY 14203
Center Director
Gene Morse, PharmD, FCCP, BCPS
- 9 of 92

The Perelman School of Medicine of the University of Pennsylvania - COE
3400 Civic Center Boulevard
Building 421, Philadelphia, PA 19104
Center Director
Susan Weiss, PhD
- 10 of 92

University of Pittsburgh Cancer Institute - COE
5117 Centre Avenue
Pittsburgh, PA 15232
Center Director
Patrick S. Moore, MD, MPH
- 11 of 92

Lerner Research Institute at Cleveland Clinic - COE
9500 Euclid Ave NB21, Cleveland, OH 44195
Center Director
Michaela Gack, PhD
- 12 of 92
University of Michigan (Division of Infectious Diseases) - COE
1500 E Medical Center Dr, #3, Ann Arbor, MI 48109
Center Directors
David Markovitz, MD
Mark Kaplan, MD
- 13 of 92

The Pathogen Research Centre (PaRC) at Western University - COE
1151 Richmond St, London, ON N6A 3K7, Canada
Center Directors
Eric J. Arts, PhD
Richard Gibson, MSc
Miguel E. Quiñones-Mateu, PhD
- 14 of 92

University of Wisconsin–Madison (UW–Madison) Global Health Institute - COE
1656 Linden Drive, Madison, WI 53706
Center Directors
Tony Goldberg, PhD
Yoshihiro Kawaoka, PhD
- 15 of 92
 Abbott Laboratories - Corporate Partner
Abbott Laboratories - Corporate Partner
100 Abbott Park Rd, Abbott Park, IL 60064, USA
Center Director
Gavin Cloherty, PhD
- 16 of 92

University of Nebraska Medical Center (UNMC) - COE
42nd and Emile, Omaha, NE 68198
Center Directors
Christopher Kratochvil, MD
Howard Gendelman, MD
- 17 of 92

College of Veterinary Medicine and Biological Sciences, Colorado State University - COE
1619 Campus Delivery, Colorado State University, Fort Collins, CO 80523-1619
Center Director
Ramesh Akkina, D.V.M, A.P, PhD
- 18 of 92

Gladstone Institute of Virology and Immunology (GIVI) - COE
1650 Owens Street, San Francisco, CA 94158
Center Director
Melaine Ott, MD, PhD
- 19 of 92

La Jolla Viral Network (LJVN) - COE
9420 Athena Circle, La Jolla, CA 92037
Center Director
Richard Wyatt, PhD
Erica Ollmann Saphire, PhD, MBA
- 20 of 92
J. Craig Venter Institute - COE
4120 Capricorn Lane, La Jolla, CA 92037
Center Director
John I. Glass, PhD
- 21 of 92
Scripps Research - COE
10550 N Torrey Pines Rd, La Jolla, CA 92037
Center Director
Richard Wyatt, PhD
- 22 of 92

University of Arizona – Aegis Consortium - COE
Health Sciences Innovation Building, 1670 East Drachman Street, Tucson, AZ 85721
Center Directors
Janko Nikolich-Žugich, MD, PhD
Jim Buizer
- 23 of 92

University Texas Medical Branch (UTMB) - COE
301 University Blvd., Galveston, TX 77550
Center Director
Scott Weaver, PhD
- 24 of 92
Tulane University School of Medicine - COE
1430 Tulane Ave, New Orleans, LA 70112
Center Director
Robert F Garry, PhD
- 25 of 92

Emory University - COE
Laboratory of Biochemical Pharmacology, 1760 Haygood Drive, Room E420, Atlanta, GA 30322
Center Directors
Raymond F. Schinazi, PhD, DSc
Carlos Del Rio, MD
- 26 of 92

University of Florida College of Medicine - COE
1600 SW Archer Rd, Gainesville, FL 32610
Center Director
Marco Salemi, PhD
- 27 of 92

University of Miami Center for AIDS Research (Miami CFAR) - COE
BCRI Building, 1580 N.W. 10th Ave., Seventh floor, Miami, FL 33136 USA
Center Director
Mario Stevenson, PhD
- 28 of 92

University of the West Indies at St. Augustine in Trinidad and Tobago - Affiliate
The University of the West Indies, St. Augustine, Trinidad, W.I.
Center Director
Christine V. F. Carrington, PhD
- 29 of 92

University of the West Indies Mona - Affiliate
Kingston 7
Jamaica, W.I.
Center Director
Joshua James Anzinger, PhD
- 30 of 92
Consortium
Colombia-Wisconsin One-Health Consortium (CWOHC)
Carrera 87 N° 30 65, Medellin, Colombia
Center Director
Jorge Ozorio, D.V.M, PhD
- 31 of 92

The Tropical Medicine Institute “Alexander von Humboldt” of the Peruvian University Cayetano Heredia - COE
Universidad Peruana Cayetano Heredia, Av. Honorio Delgado 430, San Martín de Porres, Peru
Center Director
Eduardo Gotuzzo, MD
Theresa J. Ochoa, MD, PhD
- 32 of 92

Institute of Biotechnology & Molecular Biology (IBBM) La Plata National University - COE
Universidad Nacional de La Plata, Calle 49 y 115 1900 La Plata, Argentina
Center Directors
Victor Romanowski, PhD
Mariana Viegas, PhD
- 33 of 92

Institut Pasteur São Paulo (Scientific Platform Pasteur-USP) - COE
Av. Prof. Lucio Martins Rodrigues, 370, Bloco A, 3° e 4° andares – Butantã, São Paulo – Brasil – CEP: 05508-020
Center Directors
Luís Carlos S. Ferreira, PhD
Paola Minoprio, PhD
Rubens Alves, PhD
- 34 of 92

The Department of Microbiology, Immunology and Parasitology at the Federal University of São Paulo - COE
Rua Botucatu, 862
Vila Clementino - São Paulo - SP
CEP: 04023-001
Center Director:
Luiz Mário Ramos Janini, MD, PhD
- 35 of 92

FIOCRUZ - Laboratory of Clinical Research in Neuroinfections (Chagas Center) - COE
Avenida Brasil, 4365, Manguinhos Rio de Janeiro, RJ 22640-361, Brazil
Center Director
Abelardo Araujo, PhD
- 36 of 92

Universidade Federal de Minas Gerais (UFMG) - COE
Av. Pres. Antônio Carlos, 6627 - Pampulha, Belo Horizonte - MG, 31270-901, Brazil
Center Director
Mauro Teixeira, PhD
PhD
- 37 of 92
St. George’s University, Grenada, West Indies - Affiliate
University Centre Grenada, West Indies, Grenada
Center Director
Calum N.L. Macpherson, PhD, DIC
- 38 of 92
 The Peter Doherty Institute of Infection and Immunity - COE
The Peter Doherty Institute of Infection and Immunity - COE
792 Elizabeth Street, Melbourne VIC 3000 Australia
Center Directors
Sharon Lewin, FRACP, PhD, FAAHMS
Damian Purcell, PhD
Peter Revill, PhD
- 39 of 92
Griffith University, The Center for Emerging Viruses, Inflammation and Therapeutics - COE
Gold Coast Campus, Parklands Drive, Southport, Gold Coast, 4222, Queensland, Australia
Center Director
Suresh Mahalingam, PhD
- 40 of 92
Singapore Consortium
Duke-NUS Medical School
8 College Rd, Singapore 169857, Singapore
Center Director - Linfa (Lin-Fa) Wang, PhD FTSE
Defence Science Organisation (DSO) National Laboratories
12 Science Park Drive, Singapore, 118225, Singapore
Center Director - Boon Huan Tan, PhD
Environmental Health Institute (EHI) - National Environmental Agency
40 Scotts Road, #13 - 00 Environment Building Singapore 228231
Center Director - Lee Ching Ng, PhD
Lee Kong Chian Medical School – Nanyang Technological University
Novena Campus
Headquarters & Clinical Sciences Building 11 Mandalay Road
Singapore 308232
Center Director - Dahai Luo, PhD
National Public Health Laboratory – Ministry of Health Singapore
9 Hospital Drive, Block C Level 4, 169612 Singapore
Center Director - Raymond Lin, MBBS, FRCPA
National University of Singapore - Department of Microbiology and Immunology
10 Medical Dr, Singapore 117597
Center Director - Justin Chu, PhD
Singapore Immunology Network (SIgN), A*STAR
8A Biomedical Grove, Levels 3 & 4, Immunos Building, Singapore 138648
Center Director - Lisa Ng, PhD
- 41 of 92

Mahidol Virus Network - COE
Mahidol University, 272 Rama VI Road, Ratchathewi, Bangkok 10400, Thailand
Center Director
Prasert Auewarakul, MD
- 42 of 92

Vietnamese National Institute of Hygiene and Epidemiology (NIHE), Laboratory of Molecular Diagnostics - Affiliate
1 P. Yec Xanh, Phạm Đình Hổ, Hai Bà Trưng, Hà Nội 100000, Vietnam
Center Director
Lan Anh Nguyen, MD, PhD
- 43 of 92
![]()
Wuhan Institute of Virology, Chinese Academy of Sciences - COE
44 Xiao Hong Shan, Wuhan Hubei, 430071, China
Center Director
Fei Deng, PhD
- 44 of 92

Institute for Viral Disease Control & Prevention, National Centre for AIDS/STD Control & Prevention, China CDC - COE
155 Changbai Road, Changping District, Beijing 102206, China
Center Directors
Yi Zeng, MD
Xiao-Ping Dong, PhD
- 45 of 92
Institut Pasteur Korea (IPK) - COE
16, Daewangpangyo-ro 712 beon-gil, Bundang-gu, Seongnam-si, Gyeonggi-do, 13488, Republic of Korea
Center Director
Jiho Kim, PhD
- 46 of 92

Research Center for Zoonosis Control - COE
Hokkaido University, North 20, West 10 Kita-ku, Sapporo 001-0020, Japan
Center Director
Hirofumi Sawa, MD, PhD
- 47 of 92
 National Institute of Infectious Diseases - COE
National Institute of Infectious Diseases - COE
Toyama 1-23-1, Shinjuku Tokyo 162-8640, Japan
Center Director
Hideki Hasegawa, PhD
- 48 of 92
Institute of Medical Science, University of Tokyo - COE
4-6-1 Shirokanedai Minato-ku, Tokyo 108-8639
Center Director
Yasushi Kawaguchi, PhD
- 49 of 92

Institue for Virus Research, Kyoto University - COE
53 Shogoin Kawara-cho Sakyo-ku, Kyoto 606-8507
Center Director
Yoshio Koyanagi, PhD
- 50 of 92
 Institute of Advanced Virology, Trivandrum, Kerala, India - Affiliate
Institute of Advanced Virology, Trivandrum, Kerala, India - Affiliate
JRQV+MQ9, Thonnakkal, Kerala 695317, India
Center Director
Akhil C. Banerjea, PhD
- 51 of 92

Amrita Institute of Medical Sciences and Research Center - COE
Ponekkara, Kochi, Kerala 682 041, India
Center Director
Prem Nair, MD
- 52 of 92
 Manipal Institute of Virology (MIV), Manipal Academy of Higher Education - COE
Manipal Institute of Virology (MIV), Manipal Academy of Higher Education - COE
Manipal, 576 104 Karnataka State, India
Center Director
Chrianjay Mukhopadhyay, MD, MBBS
- 53 of 92
Research Institute of Virology Ministry of Health of the Republic of Uzbekistan - Affiliate
Yangishahar Street, 7A, Tashkent, 100194, Uzbekistan
Center Director
Musabaev Erkin Isakovich, PhD
- 54 of 92

Turkish Virology Consortium (TVC) - COE
İzmir Tinaztepe University, Galen Research Center, Aydoğdu mahallesi, 1267/1. Sokak, No:4, 35400 Buca/İzmir, Turkey
Center Director
Mehmet Öztürk, PhD
- 55 of 92

Tel Aviv University Center for Nanoscience and Nanotechnology - COE
Cohen Porter United Kingdom Bldg. Rm. 229, Tel Aviv 69978, Israel
Center Director
Jonathan Gershoni, PhD
- 56 of 92
 Uganda Virus Research Institute - COE
Uganda Virus Research Institute - COE
Plot No: 51 -59 Nakiwogo Road, P.O.Box 49 Entebbe, Uganda
Center Director
Pontiano Kaleebu, MD, PhD
- 57 of 92

Africa Center of Excellence for Infectious Diseases of Humans and Animals (ACEIDHA) at the University of Zambia School of Veterinary Medicine - COE
School of Veterinary Medicine, University of Zambia, P.O. Box 32379 Lusaka, Zambia
Center Director
Benard Mudenda Hang'ombe, PhD, MSc, BVM
- 58 of 92

Antiviral Pharmacology Laboratory and Clinical Trials Research Center Virology Program at the University of Zimbabwe - Affiliate
630 Churchill Ave, Harare, Zimbabwe
Center Director
Charles Chiedza Maponga, PharmD, MHPE
Justen Manasa, PhD
- 59 of 92
 CAPRISA (Centre for the AIDS Programme of Research in South Africa) - COE
CAPRISA (Centre for the AIDS Programme of Research in South Africa) - COE
719 Umbilo Rd. Nelson R. Mandela School of Medicine, University of KwaZulu-Natal Durban, South Africa
Center Director
Salim Abdool Karim, MB, ChB
- 60 of 92

NHLS Johannesburg National Institute for Communicable Diseases (NICD) - COE
1 Modderfontein Road, Sandringham, 2131, South Africa
Center Director
Penny Moore, PhD
- 61 of 92

Institute for Human Virology - Nigeria - COE
Pent House, Maina Court, Plot 252, Herbert Macaulay Way, Central Business District P.O. Box 9396
Center Directors
Alash'le Abimiku, PhD, M.Sc
Patrick Dakum, Dip. Theo, MBBS, MPH
- 62 of 92

West African Centre for Cell Biology of Infectious Pathogens (WACCBIP), University of Ghana - COE
College of Basic and Applied Sciences, Volta Road, University of Ghana, P.O. Box LG 54, Legon, Accra, Ghana
Center Directors
Osbourne Quaye, PhD
Gordon Awandare, PhD
- 63 of 92

IRESSEF (Institut de Recherche en Santé, de Surveillance Epidémiologique et de Formation) - COE
Arrondissement 4, Rue 2D1, Pole Urbain de Diamniadio, BP 7325, Dakar 20000, Senegal
Center Director
Souleymane Mboup, PhD
- 64 of 92

Centro de Biología Molecular Severo Ochoa - COE
C. Nicolás Cabrera, 1, Fuencarral-El Pardo, 28049 Madrid, Spain
Center Director
José Felix de Celis, PhD
Esteban Domingo, PhD
- 65 of 92

Centre De Recerca En Sanitat Animalas (CReSA) at the Universitat Autonoma de Barcelona (UAB) - COE
IRTA Torre Marimon, 08140 Caldes de Montbui, Barcelona
Center Directors
Joaquim Segalés, PhD
Natàlia Majó,PhD
- 66 of 92

8 Avenue Saint-Martin, 98000 Monaco
Center Directors
Patrick Rampal, PhD
Thomas Althaus, PhD
- 67 of 92
Italy Consortium
Istituto Nazionale Tumori “Fondazione Pascale” National Cancer Institute, Naples
Via Mariano Semmola, 80131 Naples, Italy
Center Director- Franco Maria Buonaguro, PhD
Istituto Zooprofilattico Sperimentale delle Venezie (IZSVe)
Viale dell'Università 10, 35020 Legnaro (PD), Italy
Center Directors - Giovanni Cattoli, PhD, Calogero Terregino, Paola de Benedictis, Isabella Monne
Instituto Zooprofilattico Sperimentale dell'Umbria e delle Marche (IZSUM)
Via Gaetano Salvemini, 1, 06126 Perugia PG, Italy
Center Director - Maria Serena Beato, PhD
San Raffaele University and Research Hospital
Via Olgettina, 60, 20132 Milan, Italy
Center Director - Guido Poli, MD
Università degli Studi dell’Insubria
Via Ravasi 2, Varese 21100
Center Director - Roberto Accolla, MD, PhD
University of Brescia
Piazza del Mercato, 15 - 25121 Brescia
Center Director - Arnaldo Caruso, PhD
National Institute of Infectious Diseases Lazzaro Spallanzani
Via Portuense, 292, 00149 Rome, Italy
Center Director - Giuseppe Ippolito, MD
University of Padova Department of Surgery, Oncology & Gastroenterology
via Gattamelata 64, 35128 Padua
Center Directors - Maria Luisa Calabro, Vincenzo Ciminale, Donna Mia D'Agostino, Annarosa del Mistro, Anita DeRossi, Antonio Rosato, Paola Zanovello
- 68 of 92
 Institute of Virology, Technical University of Munich/Helmholtz Center Munich - COE
Institute of Virology, Technical University of Munich/Helmholtz Center Munich - COE
Trogerstrasse 30, 81675 Munich, Germany
Center Directors
Ulrike Protzer, MD, PhD
Oliver Keppler, MD, PhD
Michael Hoelscher, MD, PhD
- 69 of 92
Fondation Mérieux - COE
17, rue Bourgelat 69002 Lyon - France
Center Director
Marc Bonneville, DVM, PhD
- 70 of 92

Institut Pasteur - COE
25-28 Rue du Dr Roux, 75015 Paris, France
Center Directors
Marc Lecuit, MD, PhD
Antoine Gessain, MD, PhD
- 71 of 92
The Pirbright Institute - COE
Pirbright Laboratory, Ash Road, Pirbright, Surrey, England GU24 0NF, UK
Center Director
Bryan Charleston, MRCVS, FRS
- 72 of 92
University College Dublin - COE
Belfield, Dublin 4, Ireland
Center Director
William Hall, PhD
- 73 of 92

MRC-University of Glasgow Centre for Virus Research - COE
Sir Michael Stoker Building
Garscube Campus
464 Bearsden Road
Glasgow G61 1QH
Scotland (UK)
Center Directors
Massimo Palmarini, DVM, PhD
Emma Thomson, PhD
- 74 of 92

Rega Institute, KU Leuven - COE
Herestraat 49 - box 1040
3000 Leuven Belgium
Center Director
Johan Neyts, PhD
- 75 of 92
Department of Virology, Erasmus University Medical Center - COE
Dr. Molewaterplein 40, 3015 GD Rotterdam, The Netherlands
Center Director
Marion Koopmans, PhD, DVM
- 76 of 92
Radboud University Medical Center - COE
Geert Grooteplein Zuid 10, 6525 GA Nijmegen, The Netherlands
Center Director
Mihai Netea, PhD
- 77 of 92

Institute for Virology, Philipps-Universität Marburg - COE
Hans-Meerwein Straße 2
35043 Marburg, Germany
Center Director
Stephan Becker, PhD
- 78 of 92

University of Southern Denmark - COE
Campusvej 55, 5230 Odense M, Denmark
Center Director
Christine Benn, PhD
- 79 of 92
University of Veterinary Medicine Hannover - COE
Stiftung Tierärztliche Hochschule Hannover
Bünteweg 2
30559 Hannover
Center Director
Ab Osterhaus, DVM PhD
- 80 of 92
Robert Koch Institute - COE
Nordufer 20, 13353 Berlin, Germany
Center Director
Lothar H. Wieler, PhD
- 81 of 92
Karolinska Institute - COE
Department of Laboratory Medicine, Alfred Nobels Allé 8, SE-17177 Flemingsberg, Sweden
Center Director
Anders Vahlne, MD, PhD
- 82 of 92

University of Tartu - Affiliate
Ülikooli 18, 50090 Tartu, Estonia
Center Director
Andres Merits, PhD
- 83 of 92

Smorodintsev Research Institute of Influenza (the Institute) - COE
15/17 Ulitsa Prof. Popova, St. Petersburg, 197376, Russia
Center Director
Dmitry A. Lioznov, MD
Daria M Danilenko, PhD
- 84 of 92
Institute of Biomedical Systems and Biotechnology at the Peter the Great St. Petersburg Polytechnic University - COE
Polytechnicheskaya St, 29, Saint Petersburg, Russia, 195251
Center Director
Dimitry Moshkoff, MD
Daria M Danilenko, PhD
- 85 of 92
Moscow Center for HIV/AIDS Prevention and Treatment - COE
8th Sokolinoy gory str. 15, korp. 5, Moscow, Russian Federation, 105275
Center Director
Alexey Mazus, DMSc
- 86 of 92

MP Chumakov Center for Research and Development of Immunobiological Preparations - COE
Premises 8, bldg. 1, Village of Institute of Poliomyelitis, Settlement "Moskovskiy", Moscow, 108819, Russia
Center Director
Ishmukhammetov Aidar Airat, DMSc
- 87 of 92
Gembloux Agro-Bio Tech - COE
University of Liège, Passage des Déportés, 2
B-5030 Gembloux
Belgium
Center Directors
Arséne Burny MD, PhD
Luc Willems, PhD
- 88 of 92
Duke University School of Medicine - COE
40 Duke Medicine Circle, 124 Davison Building, Durham, NC 27710
Center Directors
Thomas Denny, PhD
Kevin Saunders, PhD
- 89 of 92

The National Institute of Virology and Bacteriology (NIVB) - COE
Flemingovo náměstí 542/2, 160 00 Prague 6, Czechia (Czech Republic)
Center Director
Zdeněk Hostomský, PhD
- 90 of 92

Australian Infectious Diseases Research Centre - COE
Institute for Molecular Biosciences
St Lucia 4067 QLD
Australia
Center Director
Daniel Watterson, PhD
- 91 of 92
Consortium
Sun Yat-sen University
190 Kai Yuan Avenue, Guangzhou Institute of Biomedicine & Health, Guangzhou 510530
Beijing University of Technology
Chaoyang, China, 100021, Beijing
Peking University
5 Yiheyuan Rd, Haidian District, Beijing, China, 100871
Wuhan University
Xiao Hong Shan No. 44, College of Life Sciences, Wuhan 430071
Fudan University
220 Handan Rd, Yangpu District, Shanghai, China, 200437
China Medical University
92 Beier Road, Shenyang, Liaoning 110001
Nankai University
94 Weijin Rd, Nankai District, Tianjin, 300071
Chinese Academy of Sciences
52 Sanlihe Rd, Xicheng District, Beijing, 100045
National Institutes for Food & Drug Control
1 Beiluyuan Zhanlan Road, Xicheng District, Beijing, 100037
Chinese Academy of Medical Sciences
16, Nanxiao street, Dongzhimen, Dongcheng District, Beijing, 100700
Fujian Centre for Disease Control & Prevention
1, Jianxing Road, Fuzhou, Fujian, 350011
Centers Director
Yiming Shao, PhD
- 92 of 92