Background
SUBVARIANT BA.1
- The first known confirmed B.1.1.529 infection was from a specimen collected on 9 November 2021.
- This variant has been detected at faster rates than previous surges in infection, suggesting that this variant may have a growth advantage.
- High numbers of spike mutations (at least 32 mutations) raise concerns.
- The variant shares similarities with the Lambda and Beta variants, which are associated with an innate evasion of immunity.
- The key mutations in the spike protein are K417N, N440K, G446S, S477N, T478K, E484A, Q493K, G496S, Q498R, N501Y, Y505H and P681H.
Reference
- WHO – Classification of Omicron (B.1.1.529): SARS-CoV-2 Variant of Concern.
WHO, November 26, 2021
SUBVARIANT BA.2
- BA.2 was first identified in India and South Africa in late December 2021.
- This emerged from a mutation of the Omicron variant (officially known as BA.1).
- Early observations from India and Denmark suggest there is no dramatic difference in severity compared to BA.1.
- BA.1 and BA.2 share 32 mutations, but differ by 28 mutations.
- BA.2 does not have deletion (Δ69-70) in the S protein, a major mutation found in BA1.
- As of January 24, 2022, BA.2 is becoming dominant in at least India, Denmark and Sweden.
Reference
WHO – Statement on Omicron sublineage BA.2
WHO, February 22, 2022
Background
SUBVARIANT BA.3
- BA.3 was originally discovered in northwestern South Africa.
- This subvariant is a combination of mutations in BA.1 and BA.2 spike proteins.
- Of the 33 mutations in the BA.3 lineage spike protein, 31 mutations are common to BA.1.
- BA.3 is currently causing the lowest number of cases in these three lineages. It may have been due to the loss of six mutations (ins214EPE, S371L, G496S, T547K, N856K, and L981F) from BA.1 or obtaining two mutations from BA.2 (S371F and D405N).
Reference
Emergence of Omicron third lineage BA.3 and its importance
Journal of Medical Virology, January 18, 2022
Efficacy of Vaccines
Neutralization of the SARS-CoV-2 Omicron BA.1 and BA.2 Variants
NEJM, March 16, 2022
- After the initial two doses of the BNT162b2 (Pfizer) vaccine, the median pseudovirus neutralizing antibody titers against WA1/2020, BA.1, and BA.2 were 658, 29, and 24, respectively.
- Six months after the initial vaccination, the median neutralizing antibody titers declined to 129 for WA1/2020 and to less than 20 for both BA.1 and BA.2.
- Two weeks after the third dose (booster) of the BNT162b2 vaccine, the median neutralizing antibody titers increased substantially to 6539 for WA1/2020, 1066 for BA.1, and 776 for BA.2, indicating that the median neutralizing antibody titer against WA1/2020 was 6.1 and 8.4 times those for BA.1 and BA.2, respectively.
Efficacy of a Fourth Dose of Covid-19 mRNA Vaccine against Omicron
NEJM, March 16, 2022
- After the fourth dose, both Pfizer and Moderna vaccines induced IgG antibodies against SARS-CoV-2 receptor-binding domain and increased neutralizing antibody titers; each measure was increased by a factor of 9 to 10, to titers that were slightly higher than those achieved after the third dose, with no significant difference between the two vaccines.
- Both vaccines induced an increase in live neutralization of the B.1.1.529 (omicron) variant and other viral strains by a factor of approximately 10, similar to the response after the third dose.
- The fourth dose did not lead to substantial adverse events despite triggering mild systemic and local symptoms in the majority of recipients
- Vaccine efficacy was estimated to be higher for the prevention of symptomatic disease (43% for BNT162b2 and 31% for mRNA-1273).
- Most of the infected participants were potentially infectious, with relatively high viral loads.
- A fourth dose of mRNA vaccine is immunogenic, safe, and somewhat efficacious (primarily against symptomatic disease).
- A comparison of the initial response to the fourth dose with the peak response to a third dose did not show substantial differences in humoral response or in levels of omicron-specific neutralizing antibodies.
Efficacy of Therapeutic Antibodies
Efficacy of Antiviral Agents against the SARS-CoV-2 Omicron Subvariant BA.2
NEJM, March 9, 2022
- Both LY-CoV016 (marketed as etesevimab) and LY-CoV555 (marketed as bamlanivimab), individually and in combination, lost neutralizing activity against omicron/BA.2 (NCD1288).
- REGN10987 (marketed as imdevimab), which was previously shown to lose neutralizing activity against omicron/BA.1 (NC928) and omicron/BA.1.1 (NC929) had neutralizing activity against omicron/BA.2 (NCD1288).
- The combination of REGN10987 and REGN10933 (marketed as casirivimab) also inhibited omicron/BA.2 but did not inhibit omicron/BA.1 or omicron/BA.1.1.
- REGN10933, COV2-2196 (marketed as tixagevimab), and COV2-2130 (marketed as cilgavimab) neutralized omicron/BA.2.
- S309 (the precursor of sotrovimab) had low neutralizing activity against omicron/BA.2.
- All together, this study showed that some therapeutic monoclonal antibodies (REGN10987–REGN10933, COV2-2196–COV2-2130, and S309) have lower neutralizing activity against omicron/BA.2 than against earlier variant strains.
- The susceptibilities of omicron/BA.2 (NCD1288) to remdesivir, molnupiravir, and nirmatrelvir were similar to those of the ancestral strain and other variants of concern.
Infection period of the Omicron variant
- Recent analysis from the UK Health Security Agency suggests that the window between infection and infectiousness may be shorter for the Omicron variant than the Delta variant.
- Data from recent outbreaks of Omicron demonstrate that the incubation period of Omicron may be as short as 3-5 days (compared to 5-7 days for the Delta variant and 7-14 days for the original SARS-CoV-2).
Reference
- Outbreak caused by the SARS-CoV-2 Omicron variant in Norway, November to December 2021
Eurosurveillance, December 16, 2021
RT-PCR assays for specific detection of the Omicron variant
- Molecular detection of SARS-CoV-2 strains and differentiation of Delta variant strains
Transbound Emerging Diseases, December 29, 2021 - Specific Detection of SARS-CoV-2 B.1.1.529 (Omicron) Variant by Four RT-qPCR Differential Assays
MedRxiv, December 9, 2021 - In Silico Design of Specific Primer Sets for the Detection of B.1.1.529 SARS-CoV-2 Variant of Concern (Omicron)
Zenodo, December 1, 2021
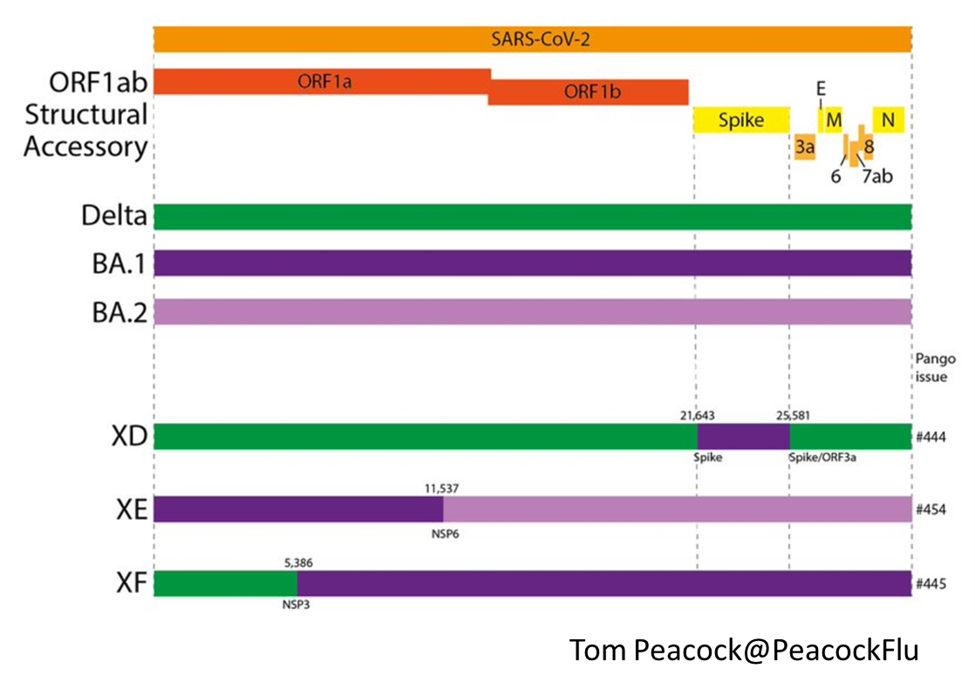
Emergence of Recombinant Variants
- Currently, There is no evidence that these recombinant variants are more transmissible or cause “more severe” outcomes.
- XD (Deltacron) emerged from the recombination of Omicron BA.1 and Delta variants.
- XE (Stealth Omicron) is the combination of Omicron BA.1 and the highly-transmissible BA.2.
- XF (Deltacron) is a UK Delta and BA.1 lineage