Ongoing Outbreaks of Lassa Virus in Nigeria
Ongoing Outbreaks of Lassa Virus in Nigeria
March 8, 2022
Context
The first known confirmed Omicron (B.1.1.529) infection was from a specimen collected on November 9, 2021 in Botswana (1). The variant rapidly began to spread to South Africa and other Southern African countries. Nigeria also experienced daily surge of COVID-19 cases in December, and Omicron became the dominant variant. Currently, cases of COVID-19 substantially are decreasing daily. However, the cases of Lassa fever are much higher than the previous epidemic seasons. This attributes to the reduced response capacity in surveillance and laboratory testing due to conversion of dedicated Lassa fever facilities into COVID-19 health-care facilities (2).
Lassa virus (LASV) is an Old World arenavirus that causes Lassa fever, an often-fatal viral hemorrhagic fever (3). It is estimated that there are approximately 100,000–300,000 clinical infections in West Africa per year, with approximately 5000 deaths (4). Case fatality estimates range from 10–69%, but can be more than 90% in the third trimester of pregnancy. Lassa fever is classified as a National Institute of Allergy and Infectious Diseases Category A Biodefense Agent (5) and is endemic to Sierra Leone, Liberia, Guinea, and Nigeria (6). Sporadic cases have been reported in other West African countries, with recent outbreaks in Togo and Benin. The natural reservoir for the virus is the Mastomys natalensis rodent (commonly known as the multimammate rat) (7). Transmission of the virus to humans occur through: (1) direct contact with urine, faces, saliva or blood of infected rats; (2) contact with objects, household items and surfaces or eating food, contaminated with urine, feces, saliva or blood of infected rats and (3) person to person transmission by contact with blood, urine, feces, vomitus, and other body fluids of an infected person (8).
Reported Cases
In Nigeria, from 3 to 30 January 2022, 211 laboratory confirmed Lassa fever cases including 40 deaths (case fatality ratio: 19%) have been cumulatively reported in 14 of the 36 Nigerian states and the Federal Capital Territory across the country (2) with majority (73%) of the infections concentrated in three states; Bauchi in the north and Edo and Ondo in the south (NCDC Lassa Fever Situation Feb 2022 report). In contrast, noticeably less Lassa fever cases with less geographical spread were reported for the same period in 2021 (54 confirmed cases, including 12 deaths, from 8 States). Nigerian health officials reported an additional 58 confirmed Lassa fever cases with the death toll of six during the first week of February (9). The annual peak of Lassa fever cases is typically observed during the dry season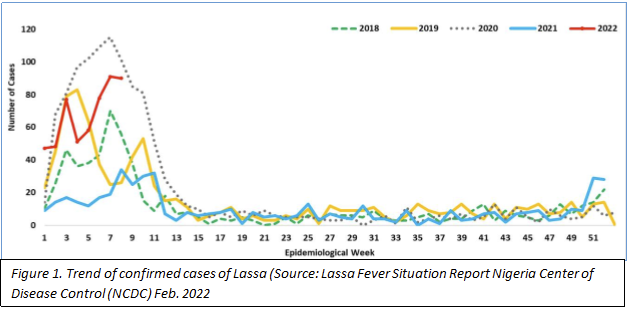
the end of the dry season as illustrated in figure 1 (2). In addition, the U.K. has had three new confirmed cases of Lassa fever with one death (10). The cases are within the same family and are linked to recent travel to West Africa
Lassa virus (LASV) is an Old World arenavirus that causes Lassa fever, an often-fatal viral hemorrhagic fever (3). It is estimated that there are approximately 100,000–300,000 clinical infections in West Africa per year, with approximately 5000 deaths (4).
In Nigeria, from 3 to 30 January 2022, 211 laboratory confirmed Lassa fever cases including 40 deaths (case fatality ratio: 19%) have been cumulatively reported in 14 of the 36 Nigerian states and the Federal Capital Territory across the country (2) with majority (73%) of the infections concentrated in three states; Bauchi in the north and Edo and Ondo in the south (NCDC Lassa Fever Situation Feb 2022 report).
Even with ongoing pandemic, our efforts must be continued for the viral surveillance of bats, pigs, and people with One Health approach and for enhancing our understanding on the ecology of Pteropus bats and the dynamics of the virus spillover event to intermediate hosts and people.
 The “Enable” study
The “Enable” study
There is no licensed Lassa fever vaccine (5). Perhaps the most significant effort toward developing an effective vaccine for Lassa is the ‘Enable’ study funded by the Coalition for Epidemic Preparedness Innovations (CEPI) that plans to enroll up to 23,000 participants across five West African countries (Benin, Guinea, Liberia, Nigeria, and Sierra Leone). The ‘Enable’ study hopes to establish important epidemiological data to include spread, risk factors, and incidence that would inform joint multi-country Lassa Fever Vaccine Efficacy and Prevention for West Africa (LEAP4WA) planned by CEPI and European & Developing Countries Clinical Trials Partnership (EDCTP) across the selected West African countries (ENABLE ; LEAP4WA). The only available treatment, ribavirin, is most effective only during early infection (11). Lassa fever is most often diagnosed by using enzyme-linked immunosorbent serologic assays (ELISA), which detect IgM and IgG antibodies as well as Lassa antigen (12). Reverse transcription-polymerase chain reaction (RT-PCR) can be used in the early stage of disease. Recently, great progresses have made for treatment and prophylaxis of Lassa fever by in-depth understanding of viral protein structure. A single surface glycoprotein complex (GPC) is responsible for attachment and entry of LASV into host cells (13). GPC is a heavily N-glycosylated trimer that is composed of three non-covalently associated subunits, GP1 and GP2 and an unusual stable signal peptide (SSP). GP1 is responsible for receptor binding, while GP2 mediates fusion of the virion with a cell membrane. SSP is required for proper processing of GPC and is retained as part of the complex. As the sole antigen on the viral surface, GPC is the primary target of neutralizing antibodies (MAbs). Neutralizing monoclonal antibodies have been isolated from Sierra Leonean and Nigerian survivors of Lassa fever (6). Development of MAbs as potential Lassa immunotherapeutics was facilitated by structural studies and mutational analyses that identified protective epitopes on the prefusion form of the LASV glycoprotein (14-16). Use of structure-guided amino acid substitutions has led to increase in the neutralization potency and breadth of these antibodies to include all major LASV lineages (16). The ability to define antibody residues that allow potent and broad neutralizing activity, together with findings from analyses of inferred germline precursors, is critical to develop potent therapeutics. Antibody therapeutics may be further developed in clinical trials in endemic areas potentially offering a key treatment option for Lassa fever. In vaccine design and assessment, vaccine-driven elicitation of anti-LASV GPC-B antibodies efficiently in humans may be possible only through immunization with an engineered immunogen that faithfully and sustainably displays the prefusion trimer of LASV GPC. Further, site-selective deglycosylation of viral glycoproteins can improve immunogenicity and may be also an effective strategy for the extensively glycan-shielded Lassa virus, particularly through the incorporation of N-glycan deletions at positions 390 and 395 in the GPC (16).
 Conclusion
Conclusion
LASV infection poses a significant healthcare burden in those living in rural areas where communities have poor sanitation and/or crowded living conditions. The major control strategy is the control of the rodents around dwellings, avoiding of rats consumption and contact (17). The lack of an approved therapeutic or vaccine, potential for geographic expansion of the rodent reservoir, ease of procurement and weaponization of the virus, and the possible emergence of new viral strains support recommendations for enhanced surveillance and preparedness for Lassa fever (6). It is necessary to continue to enhance viral surveillance effort for early detection and treatment of cases to reduce the case fatality rate.
Our GVN Team thanks Dr. Alash’le Abimiku (The Institute of Human Virology, University of Maryland) for reviewing this manuscript and for providing us with invaluable information, comments, and insights.
References
- World Health Organization. Update on Omicron. https://www.who.int/news/item/28-11-2021-update-on-omicron.
- World Health Organization. Lassa Fever – Nigeria. https://www.who.int/emergencies/disease-outbreak-news/item/lassa-fever—nigeria.
- Shaffer JG, Grant DS, Schieffelin JS, Boisen ML, Goba A, Hartnett JN, Levy DC, Yenni RE, Moses LM, Fullah M, Momoh M, Fonnie M, Fonnie R, Kanneh L, Koroma VJ, Kargbo K, Ottomassathien D, Muncy IJ, Jones AB, Illick MM, Kulakosky PC, Haislip AM, Bishop CM, Elliot DH, Brown BL, Zhu H, Hastie KM, Andersen KG, Gire SK, Tabrizi S, Tariyal R, Stremlau M, Matschiner A, Sampey DB, Spence JS, Cross RW, Geisbert JB, Folarin OA, Happi CT, Pitts KR, Geske FJ, Geisbert TW, Saphire EO, Robinson JE, Wilson RB, Sabeti PC, Henderson LA, Khan SH, Bausch DG, Branco LM, Garry RF, Viral Hemorrhagic Fever Consortium. PLoS Negl Trop Dis. 2014 Mar; 8(3):e2748.
- McCormick JB, Webb PA, Krebs JW, Johnson KM, Smith ES. A prospective study of the epidemiology and ecology of Lassa fever. J Infect Dis. 1987, 155(3):437-44.
- National Institute of Allergy and Infectious Diseases NIAID Emerging Infectious Diseases/Pathogens. 2018. https://www.niaid.nih.gov/research/emerging-infectious-diseases-pathogens.
- Garry R.F. (2020) 50 Years of Lassa Fever Research. In: Current Topics in Microbiology and Immunology. Springer, Berlin, Heidelberg.
- Monath TP, Newhouse VF, Kemp GE, Setzer HW, Cacciapuoti A. Lassa virus isolation from Mastomys natalensis rodents during an epidemic in Sierra Leone. Science. 1974 Jul 19; 185(4147):263-5.
- Yaro, C.A., Kogi, E., Opara, K.N. et al. Infection pattern, case fatality rate and spread of Lassa virus in Nigeria. BMC Infect Dis 21, 149 (2021).
- Outbreak News Today. Nigeria reports additional 58 confirmed Lassa fever cases in first week of February. http://outbreaknewstoday.com/nigeria-reports-additional-58-confirmed-lassa-fever-cases-in-first-week-of-february-80471/
- Forbes. Lassa Fever Leaves One Dead, 2 Others Infected In United Kingdom. February 13, 2022. https://www.forbes.com/sites/brucelee/2022/02/13/lassa-fever-leaves-one-dead-2-others-ill-in-united-kingdom/?sh=7b80eabc2bf0
- McCormick JB, King IJ, Webb PA, Scribner CL, Craven RB, Johnson KM, Elliott LH, Belmont-Williams R. Lassa fever. Effective therapy with ribavirin. N Engl J Med. 1986 Jan 2; 314(1):20-6.
- CDC. Lassa Fever-Diagnosis. https://www.cdc.gov/vhf/lassa/diagnosis/index.html#:~:text=Lassa%20fever%20is%20most%20often,the%20early%20stage%20of%20disease.
- Messina EL, York J, Nunberg JH. Dissection of the role of the stable signal peptide of the arenavirus envelope glycoprotein in membrane fusion. J Virol. 2012 Jun; 86(11):6138-45.
- Robinson JE, Hastie KM, Cross RW, Yenni RE, Elliott DH, Rouelle JA, Kannadka CB, Smira AA, Garry CE, Bradley BT, Yu H, Shaffer JG, Boisen ML, Hartnett JN, Zandonatti MA, Rowland MM, Heinrich ML, Martínez-Sobrido L, Cheng B, de la Torre JC, Andersen KG, Goba A, Momoh M, Fullah M, Gbakie M, Kanneh L, Koroma VJ, Fonnie R, Jalloh SC, Kargbo B, Vandi MA, Gbetuwa M, Ikponmwosa O, Asogun DA, Okokhere PO, Follarin OA, Schieffelin JS, Pitts KR, Geisbert JB, Kulakoski PC, Wilson RB, Happi CT, Sabeti PC, Gevao SM, Khan SH, Grant DS, Geisbert TW, Saphire EO, Branco LM, Garry RF. Most neutralizing human monoclonal antibodies target novel epitopes requiring both Lassa virus glycoprotein subunits. Nat Commun. 2016 May 10;7:11544.
- Cross RW, Hastie KM, Mire CE, Robinson JE, Geisbert TW, Branco LM, Ollmann Saphire E, Garry RF. Antibody therapy for Lassa fever. Curr Opin Virol. 2019, 37:97-104. doi: 10.1016/j.coviro.2019.07.003.
- Hastie KM, Cross RW, Harkins SS, Zandonatti MA, Koval AP, Heinrich ML, Rowland MM, Robinson JE, Geisbert TW, Garry RF, Branco LM, Saphire EO. Convergent Structures Illuminate Features for Germline Antibody Binding and Pan-Lassa Virus Neutralization. Cell. 2019 Aug 8;178(4):1004-1015.e14. doi: 10.1016/j.cell.2019.07.020. PMID: 31398326; PMCID: PMC6814208.
- Lo Iacono G, Cunningham AA, Fichet-Calvet E, Garry RF, Grant DS, Khan SH, Leach M, Moses LM, Schieffelin JS, Shaffer JG, Webb CT, Wood JL. Using modelling to disentangle the relative contributions of zoonotic and anthroponotic transmission: the case of lassa fever. PLoS Negl Trop Dis. 2015 Jan; 9(1):e3398.